Introduction
Are you struggling with weight loss and looking for an effective solution? Tirzepatid might be the answer you've been searching for.
Tirzepatid is an FDA-approved, once-weekly injectable medication designed to aid in weight loss and weight management. It works by addressing key factors involved in weight gain, such as appetite, fullness, and gastric emptying.
In this article, we will discuss how tirzepatide helps with weight loss and what you can expect from this innovative treatment.
What Is Tirzepatid and How Does It Work for Weight Loss?
Understanding Tirzepatid's Mechanism
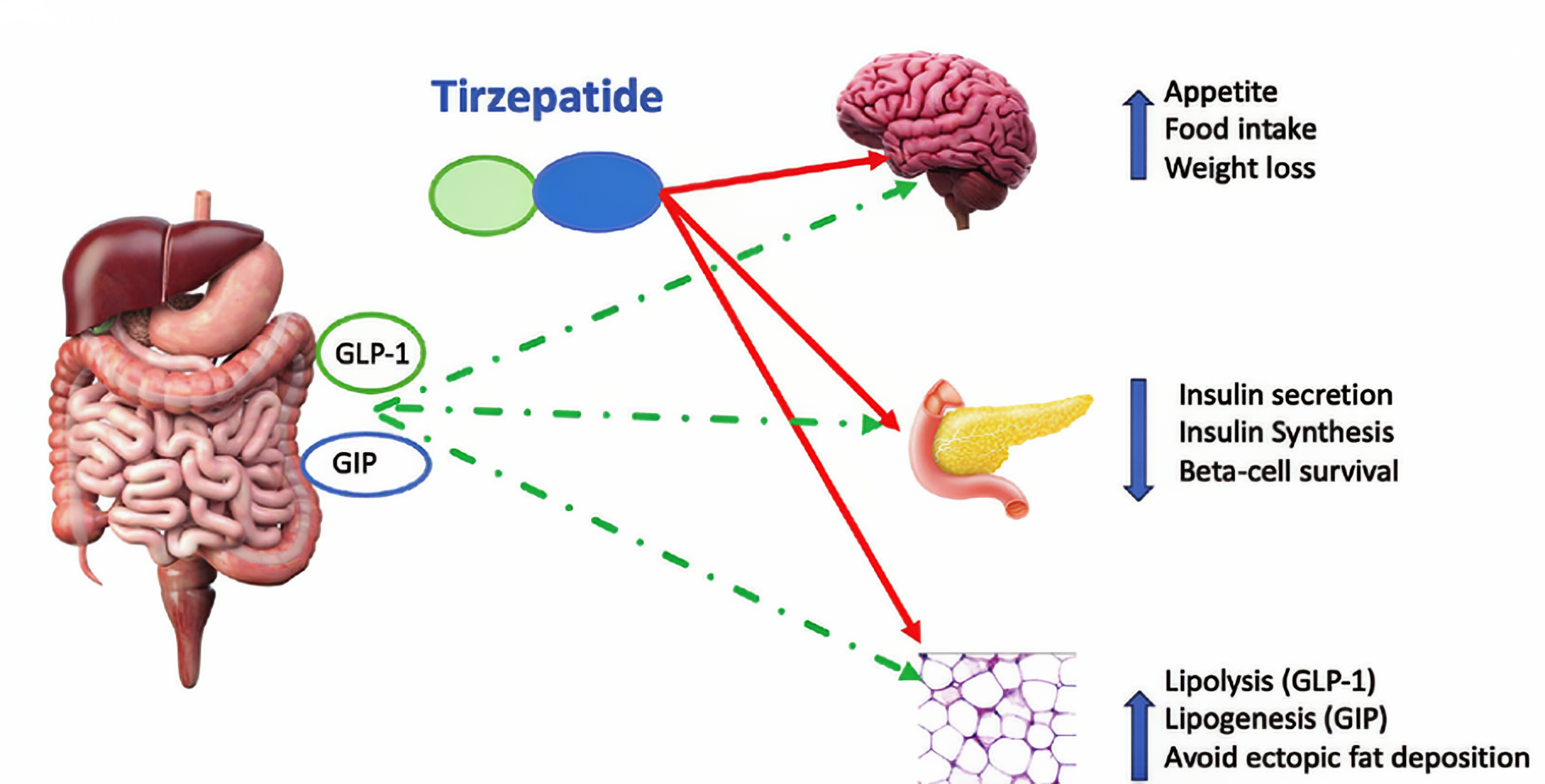
Tirzepatid is a first-in-class dual receptor agonist that targets both GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide) receptors. These two gut-derived hormones are normally released after eating, where they help regulate blood sugar, hunger, and fullness. By mimicking these natural signals, tirzepatide reduces appetite, enhances satiety, and slows digestion, creating an ideal internal environment for steady and sustainable weight loss.
Dual Action: How Tirzepatid Mimics the GLP-1 and GIP Hormones to Aid Weight Loss
What makes tirzepatide unique is its dual mechanism of action. GLP-1 has long been known to curb appetite and improve glucose control. GIP, once underestimated, has now been shown to enhance insulin sensitivity and amplify the fullness effect of GLP-1. When combined, these two pathways create a synergistic effect—stronger appetite reduction, improved metabolic balance, and greater fat-burning potential than single-receptor medications. This explains why tirzepatide often outperforms older GLP-1-only treatments.
Appetite Control and Satiety: The Role of Tirzepatid in Suppressing Appetite
Tirzepatid helps people feel full sooner and stay full longer. It works on appetite-regulating centers in the brain, lowering hunger signals and reducing cravings throughout the day. This makes it easier for individuals to naturally decrease their calorie intake without feeling deprived. Over time, this consistent reduction in daily calories leads to meaningful weight loss.
Slowing Gastric Emptying: How Delayed Stomach Emptying Helps with Weight Loss
By slowing the rate at which food leaves the stomach, tirzepatide extends the feeling of fullness after meals. This delayed gastric emptying helps reduce snacking, stabilizes blood sugar spikes after eating, and supports portion control. The result is a smoother, more controlled digestive process that aligns with healthier eating habits and long-term weight reduction.
The Science Behind Tirzepatid's Effectiveness
Dual Receptor Activation: GIP and GLP-1 Receptors' Roles in Glucose Control and Fat Loss
Tirzepatid’s ability to activate both GLP-1 and GIP receptors provides multiple metabolic advantages. GLP-1 helps regulate blood sugar, slows digestion, and reduces appetite. GIP enhances insulin sensitivity and supports fat metabolism. Together, these pathways create a powerful metabolic shift that promotes fat loss rather than muscle loss—making tirzepatide an advanced option for medically supervised weight reduction.
Increased Insulin Sensitivity: How It Supports Better Blood Sugar Regulation
Many individuals with obesity struggle with insulin resistance. Tirzepatid directly improves insulin sensitivity, allowing the body to use glucose more efficiently. This not only lowers blood sugar levels but also decreases fat storage and reduces the metabolic strain associated with obesity. For people with both obesity and type 2 diabetes, tirzepatide delivers a dual benefit: clinically significant weight loss and improved metabolic health.
Who Can Benefit from Tirzepatid for Weight Loss?
Approved Use for Obesity and Weight-Related Conditions
Tirzepatid is FDA-approved for weight loss in adults with a BMI of 30 or higher, or those with a BMI of 27 or higher who have weight-related health conditions such as hypertension, diabetes, or obstructive sleep apnea (OSA). The following table summarizes its approved uses:
Condition | BMI Requirement | Additional Conditions |
Obesity | BMI ≥ 30 | No additional conditions required |
Overweight with Health Conditions | BMI ≥ 27 | Conditions like hypertension, diabetes |
Severe OSA in Obese Adults | BMI ≥ 30 |
|
Additional Health Benefits: The Impact on Conditions Like Obstructive Sleep Apnea (OSA)
Tirzepatid not only aids in weight loss but also helps improve health conditions like OSA, which are often aggravated by obesity. By reducing weight, patients may experience improved sleep quality and overall health.
Who Should Avoid Tirzepatid?
Tirzepatid is not suitable for individuals with type 1 diabetes or a history of pancreatitis. It is also contraindicated for individuals with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN 2).
Exclusion Criteria | Reason |
Type 1 Diabetes | Not approved for use in type 1 diabetes |
History of Pancreatitis | Risk of pancreatitis may increase |
MTC or MEN 2 | Increased risk of thyroid tumors |
What Results Can You Expect from Tirzepatid for Weight Loss?
Average Weight Loss Results
Tirzepatid has shown impressive results in clinical trials. The SURMOUNT-1 trial demonstrated that patients using tirzepatide (15 mg) for 72 weeks lost an average of 22.5% of their initial body weight. Below are the results based on different dosages:
Zepbound Dose | % Weight Loss at 72 Weeks | Avg. Weight Loss (lbs) | Avg. Weight Loss (kg) |
5 mg/week | 16% | 35.5 lbs | 16.1 kg |
10 mg/week | 21.4% | 48.9 lbs | 22 kg |
15 mg/week | 22.5% | 52 lbs | 23.6 kg |
Placebo | 2.4% | 5.3 lbs | 2.4 kg |
Long-Term Results: Sustained Weight Loss in the Short-Term and Challenges in Long-Term Weight Maintenance
While tirzepatide provides substantial weight loss over the first few months, clinical trials have shown that stopping the medication may lead to weight regain. This suggests that continuous use is necessary to maintain long-term results.
Comparing Tirzepatid with Other Weight Loss Drugs
Tirzepatid vs. Semaglutid: Head-to-Head Comparison of Tirzepatid’s Superior Weight Loss Results
In the SURMOUNT-5 trial, tirzepatide was found to be 47% more effective than semaglutide for weight loss. Tirzepatid led to an average weight loss of 50.3 lbs, compared to 33.1 lbs with semaglutide. Additionally, tirzepatide resulted in a greater reduction in waist size and had a higher average percentage of weight loss.![tirzepatide for weight loss tirzepatide for weight loss]()
How Do You Use Tirzepatid for Weight Loss?
Administration Method
Tirzepatid is administered via a subcutaneous injection once a week. It can be injected at any time of the day, with or without food. The injection sites include the thigh, abdomen, or upper arm. Rotating the injection site with each dose is recommended to prevent irritation.
Injection Area | Best Practice |
Thigh | Rotate to avoid injection site irritation |
Abdomen | Rotate regularly to minimize irritation |
Upper Arm | Ideal for self-administration |
Dosing Schedule: Recommended Starting Doses and Gradual Increase Based on Tolerance
The typical dosing schedule starts with 2.5 mg per week, gradually increasing to 5 mg, 10 mg, or 15 mg based on individual tolerance. The dose is increased every four weeks to minimize side effects like nausea.
Potential Challenges with Dosing and Injection
Side Effects Management: Discuss Common Side Effects Like Nausea and Vomiting, and How to Manage Them
Common side effects include nausea, vomiting, and diarrhea. These are more common during the initial weeks of treatment but often improve as the body adjusts. To reduce these effects, patients are encouraged to eat smaller, more frequent meals and stay hydrated.
Injection Site Rotation: Best Practices for Rotating Injection Sites to Avoid Irritation
To prevent irritation, it’s essential to rotate injection sites regularly. This helps reduce the risk of localized pain, swelling, or redness from repeated injections in the same area.
What Are the Potential Side Effects of Tirzepatid?
Common Side Effects
Many individuals experience mild gastrointestinal symptoms when starting tirzepatide. These can include nausea, diarrhea, constipation, bloating, or vomiting as the digestive system adapts to slower gastric emptying. These effects are typically temporary and often improve within a few weeks of continued use. Eating smaller meals, staying hydrated, and avoiding high-fat foods during early treatment may help reduce discomfort.
Injection Site Reactions: Localized Pain or Swelling from the Injection
Some users notice redness, itching, or mild swelling at the injection site. These reactions are not harmful and usually fade quickly. Rotating injection sites—thigh, abdomen, or upper arm—helps prevent irritation. Using proper injection techniques and allowing the medication to reach room temperature before use may also reduce discomfort.
Serious Side Effects and Precautions
Gallbladder Issues and Pancreatitis: Risks of Severe Stomach Pain and What to Watch For
In rare cases, tirzepatide may trigger gallbladder problems or pancreatitis. Warning signs include sudden, severe stomach pain, pain spreading to the back, persistent vomiting, fever, or jaundice (yellowing of the skin or eyes). Because pancreatitis can be life-threatening, any severe or persistent abdominal pain should be evaluated by a healthcare provider immediately.
Thyroid and Mental Health Warnings: The Need for Monitoring Due to Potential for Thyroid Issues or Changes in Mood
Tirzepatid carries a boxed warning regarding the potential risk of thyroid C-cell tumors. Although rare, users should monitor for symptoms such as a lump in the neck, trouble swallowing, hoarseness, or shortness of breath. Any of these signs warrant prompt medical evaluation. Mood changes may also occur in sensitive individuals. Feelings of depression, anxiety, or unusual behavioral shifts should be reported to a healthcare provider without delay.
Can You Maintain Weight Loss After Stopping Tirzepatid?
Weight Regain After Discontinuation
Many people wonder whether weight loss from tirzepatide lasts after stopping treatment. Clinical studies show that weight regain is common once the medication is discontinued. In the SURMOUNT-4 trial, participants who stopped tirzepatide regained roughly 14% of the weight they had lost, even with regular follow-up. This happens because the appetite-suppressing and metabolic effects of tirzepatide no longer support reduced calorie intake once treatment stops. These findings highlight that tirzepatide functions best as part of a long-term care strategy rather than a short-term fix.
Chronic Management of Obesity: Tirzepatid as a Long-Term Solution for Weight Management
Obesity is now recognized as a chronic, relapsing condition—much like hypertension or diabetes. That means ongoing management is essential. Tirzepatid provides metabolic support that helps people maintain lower hunger levels, better glucose control, and healthier eating patterns. When used consistently, it can stabilize long-term weight management and reduce the risk of weight cycling. For many individuals, tirzepatide becomes part of a comprehensive, long-term obesity treatment plan rather than a temporary medication.
How to Avoid Weight Regain
Long-Term Use Considerations: The Importance of Maintaining Treatment for Continued Success
To maintain weight loss, patients often need to continue tirzepatide beyond the initial treatment phase. Ongoing therapy helps preserve improvements in appetite control and metabolic function. Healthcare providers may adjust dosages or recommend maintenance-level treatment depending on individual progress and side effect tolerance. Regular check-ins also help ensure that treatment remains effective and safe over time.
Supplementing with Lifestyle Changes: The Role of Diet and Exercise in Sustaining Weight Loss After Stopping Medication
Medication alone cannot guarantee permanent results. Sustainable weight maintenance requires consistent lifestyle habits. A balanced diet with manageable portions, regular physical activity, and good sleep hygiene all play vital roles in supporting long-term weight success. These habits help reinforce the progress achieved with tirzepatide and make the transition easier if the medication is reduced or discontinued later. Individuals who combine tirzepatide with healthy routines are more likely to keep weight off and avoid the rapid regain seen in clinical studies.
Conclusion
Tirzepatid offers strong support for people facing obesity or weight-related conditions. It reduces appetite, increases fullness, and helps regulate blood sugar for effective weight control. Long-term use and healthy habits may be needed to maintain results. Cocer Peptides™ provides high-quality tirzepatide solutions designed to support better outcomes and long-term weight management success.
FAQ
Q: What is tirzepatide for weight loss?
A: Tirzepatid is a weekly injection that helps reduce appetite and support weight loss.
Q: How does tirzepatide help with weight loss?
A: Tirzepatid works by increasing fullness and slowing digestion, which supports steady weight loss.
Q: Who should consider tirzepatide for weight loss?
A: Tirzepatid may help adults with obesity or weight-related conditions achieve meaningful weight loss.
Q: Is tirzepatide more effective than other weight loss drugs?
A: Tirzepatid often leads to greater weight loss compared to similar medications.
Q: What should I do if tirzepatide causes side effects?
A: Mild side effects can occur; speak with your provider if symptoms persist during tirzepatide treatment.
English
العربية
Français
Русский
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy