![network_duotone network_duotone]() By Cocer Peptides
By Cocer Peptides ![network_duotone network_duotone]() 1 month ago
1 month ago
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
In the field of life sciences, aging has always been a major research topic. As research into the mechanisms of aging continues to deepen, the role of nicotinamide adenine dinucleotide (NAD+) in the anti-aging process has garnered increasing attention. As a coenzyme involved in numerous key physiological processes within cells, NAD+ has been found to be closely linked to the aging process.
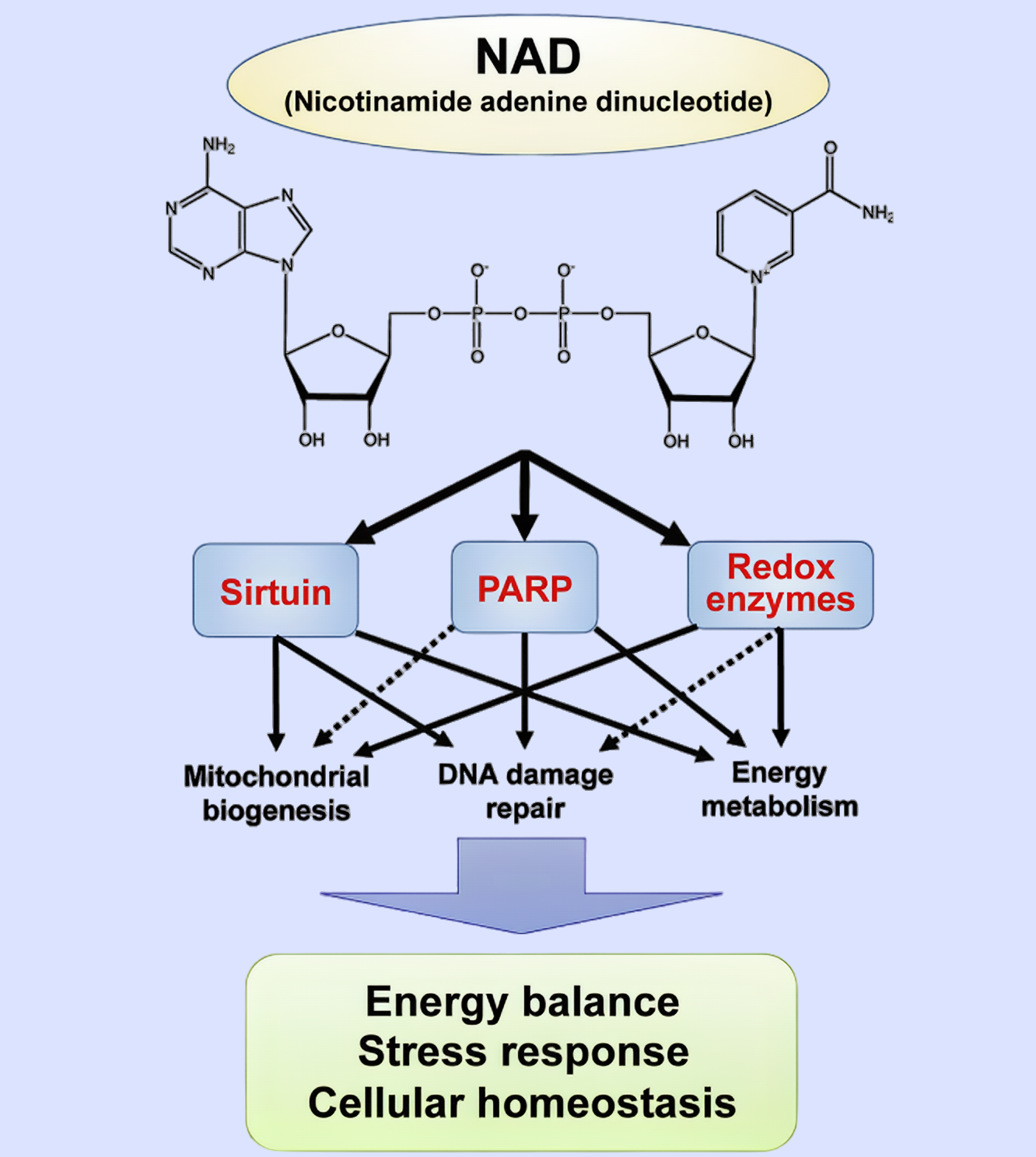
![1 1]()
Figure 1 Biological functions of NAD. NAD regulates energy balance, stress response, and cellular homeostasis through sirtuins, PARPs, and various redox enzymes.
Overview of the Physiological Functions of NAD+
NAD+ is a coenzyme widely present in cells, participating in various key physiological processes. It primarily exists in two forms within cells: the oxidized form (NAD+) and the reduced form (NADH), which can interconvert. This dynamic balance is crucial for maintaining normal cellular metabolism and function.
1. Energy metabolism: NAD+ plays a central role in cellular respiration. In energy metabolism pathways such as glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation, NAD+ acts as an electron acceptor, receiving electrons released during the oxidation of metabolic substrates to form NADH. Subsequently, NADH transfers electrons to the mitochondrial respiratory chain, where oxidative phosphorylation generates adenosine triphosphate (ATP), providing energy for the cell. This process ensures that cells can continuously obtain sufficient energy to maintain their normal physiological activities, such as cell growth, division, and repair.
During glycolysis, 3-phosphoglycerate transfers hydrogen atoms to NAD+ under the action of 3-phosphoglycerate dehydrogenase, generating NADH and 1,3-diphosphoglycerate. Subsequently, NADH transfers electrons to oxygen via the respiratory chain in the mitochondria, ultimately producing water and coupling ATP synthesis. This indicates that NAD+ is an indispensable component of cellular energy metabolism, and changes in its concentration directly affect the efficiency of energy production.
2. DNA repair: NAD+ is a substrate for the poly(ADP-ribose) polymerase (PARP) family. After PARP recognizes and binds to damaged DNA sites, it uses NAD+ as a substrate to transfer ADP-ribose groups to itself or other proteins, forming poly(ADP-ribose) (PAR) chains. These PAR chains can recruit and activate a series of proteins involved in DNA repair, such as DNA ligase and DNA polymerase, thereby initiating the DNA repair process. When cells are exposed to DNA damage caused by factors such as ultraviolet radiation or chemicals, the PARP-NAD+ system rapidly responds to repair damaged DNA and maintain genomic stability. If NAD+ levels are insufficient, PARP activity is inhibited, leading to reduced DNA repair capacity, increased genomic instability, and accelerated cellular aging and disease onset.
3. Post-translational modification of proteins: NAD+ also participates in the catalytic reactions of sirtuin family proteins. Sirtuins are a class of NAD+-dependent deacetylases that can remove acetyl modifications from lysine residues on proteins. This deacetylation modification regulates the activity, stability, and subcellular localization of numerous proteins, thereby influencing cellular metabolism, stress responses, aging, and other physiological processes. For example, SIRT1 can regulate the activity of transcription factors such as p53 and FOXO through deacetylation modification, thereby influencing cell cycle, apoptosis, and antioxidant stress processes. When cells are under stress, SIRT1 deacetylates p53 by consuming NAD+, thereby inhibiting p53's transcriptional activity, reducing the occurrence of apoptosis, and enhancing cellular survival capacity.
Changes in NAD+ Levels During Aging
Studies have shown that with age, NAD+ levels gradually decrease in multiple tissues and cells of the body. This decline has been observed in various species, including mammals, nematodes, and fruit flies, suggesting that reduced NAD+ levels may be a conserved phenomenon in the aging process.
1. Tissue-specific changes: The extent and mechanisms of NAD+ level decline with age may vary across different tissues. In skeletal muscle, aging is accompanied by a decrease in the activity of key enzymes in the NAD+ biosynthetic pathway, leading to reduced NAD+ synthesis. The expression and activity of NAD+ consuming enzymes such as CD38 increase, accelerating NAD+ degradation and ultimately resulting in a significant decrease in NAD+ levels in skeletal muscle. In the liver, in addition to the aforementioned alterations in synthesis and degradation pathways, aging may also affect NAD+ transport processes, leading to an imbalance in intracellular NAD+ distribution and further reducing its effective concentration.
2. Association with age-related diseases: Decreased NAD+ levels are closely associated with the onset and progression of various age-related diseases. In cardiovascular diseases, the decline in myocardial cell NAD+ levels caused by aging leads to energy metabolism disorders, increased oxidative stress, and myocardial cell apoptosis, thereby exacerbating cardiac dysfunction. In neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease, the reduction in neuronal NAD+ levels affects DNA repair and protein homeostasis, promoting the aggregation of neurotoxic proteins and neuronal death. Metabolic diseases such as diabetes are also associated with decreased NAD+ levels, as NAD+ deficiency impairs insulin secretion and insulin sensitivity, leading to abnormal blood glucose regulation.
Mechanisms by which decreased NAD+ levels promote aging
1. **Energy metabolism disorders**: NAD+ plays a key role in cellular energy metabolism. As age increases, reduced NAD+ levels lead to impaired energy metabolism pathways and reduced ATP production. This not only affects normal cellular physiological functions but also triggers a series of compensatory responses, such as excessive mitochondrial proliferation and functional abnormalities. Mitochondria are the cellular powerhouses; when NAD+ is insufficient, mitochondrial respiratory chain function is impaired, resulting in increased production of reactive oxygen species (ROS) during electron transport. Excessive ROS can attack mitochondrial DNA, proteins, and lipids, further disrupting mitochondrial structure and function, creating a vicious cycle that accelerates cellular aging.
![]()
![2 2]()
Figure 2 Proposed mechanisms of how aging affects NAD metabolism. Aging disrupts the balance between NAD synthesis and degradation, leading to reduced NAD levels in various tissues.
2. Accumulation of DNA damage: As a substrate for PARP, reduced NAD+ levels weaken DNA repair capacity. When DNA damage cannot be effectively repaired in a timely manner, it leads to genomic instability, accumulating a large number of mutations and chromosomal abnormalities. These genetic damages interfere with normal cellular physiological functions, affecting cell proliferation, differentiation, and apoptosis, thereby promoting cellular aging. DNA damage also activates aging-related signaling pathways within cells, such as the p53-p21 and p16INK4a-Rb pathways, further inducing the occurrence of cellular aging.
3. Dysregulation of senescence-related signaling pathways: NAD+-dependent sirtuin family proteins play a crucial role in regulating senescence-related signaling pathways. As NAD+ levels decline, sirtuin activity is inhibited, leading to reduced deacetylation modifications of downstream target proteins. Reduced SIRT1 activity results in p53 being in a highly acetylated state, enhancing p53's transcriptional activity, leading to cell cycle arrest and apoptosis; simultaneously, weakened deacetylation of the FOXO transcription factor by SIRT1 affects the cell's antioxidant stress resistance and metabolic regulation. Additionally, alterations in the activity of other sirtuin family members such as SIRT3 and SIRT6 also impact mitochondrial function, genomic stability, and inflammatory responses, collectively driving the progression of cellular senescence.
Anti-aging strategies to increase NAD+ levels
Given the close relationship between reduced NAD+ levels and aging, strategies to delay aging by increasing NAD+ levels have become a research hotspot.
1. Supplementing NAD+ Precursors: Supplementing NAD+ precursors is a common method to increase NAD+ levels. Common NAD+ precursors include nicotinamide (NAM), nicotinamide mononucleotide (NMN), and nicotinamide riboside (NR). These precursors can be converted into NAD+ through specific metabolic pathways within cells, thereby increasing its levels.
Nicotinamide (NAM): NAM is a form of vitamin B3 that can be converted into nicotinamide mononucleotide (NMN) through the action of nicotinamide phosphoribosyltransferase (NAMPT), which is then used to synthesize NAD+. High-dose NAM supplementation may feedback inhibit NAMPT activity, limiting its ability to increase NAD+ levels. Long-term high-dose use of NAM may cause side effects such as skin flushing, but at appropriate doses, NAM can effectively increase intracellular NAD+ levels, improve energy metabolism, and enhance DNA repair functions.
Nicotinamide mononucleotide (NMN): NMN is a direct precursor in the NAD+ biosynthetic pathway. Studies have shown that oral NMN is rapidly absorbed and converted into NAD+, effectively increasing NAD+ levels in various tissues. In animal experiments, NMN supplementation has shown significant improvements in age-related metabolic disorders, cardiovascular dysfunction, and neurodegenerative diseases. For example, in aged mice, NMN supplementation improved locomotor ability, enhanced insulin sensitivity, alleviated age-related pathological changes in the heart, and enhanced cognitive function. Additionally, NMN has been shown to promote mitochondrial biogenesis, enhance mitochondrial function, and reduce oxidative stress-induced damage.
Nicotinamide riboside (NR): NR is another effective NAD+ precursor that can be converted into NMN through phosphorylation by nicotinamide riboside kinase (NRK), which is then used to synthesize NAD+. Similar to NMN, supplementation with NR can increase intracellular NAD+ levels, improve metabolic function, and delay aging. In aged mice, NR supplementation can remodel metabolic and stress response pathways, enhance the chromatin-binding capacity of the circadian clock gene BMAL1, restore mitochondrial respiratory rhythms and circadian activity, and partially restore the physiological state of aged mice to that of younger mice.
![]()
![3 3]()
Figure 3 Model depicting the NAD+ salvage pathway and nicotinamide riboside (NR) conversion to NAD+.
2. Regulation of NAD+ metabolic enzymes:
Activation of NAD+ synthase: NAMPT is the rate-limiting enzyme in the NAD+ biosynthetic pathway, and increased activity can promote NAD+ synthesis. Some natural compounds, such as resveratrol and apigenin, have been found to activate NAMPT, thereby increasing NAD+ production. Resveratrol is a polyphenolic compound found in grape skins, red wine, and other plants. It can indirectly upregulate NAMPT expression by activating the SIRT1-PGC-1α signaling pathway, thereby increasing NAD+ levels. Resveratrol treatment improves energy metabolism, reduces oxidative stress damage, and extends lifespan in aged mice.
Inhibiting NAD+ consuming enzymes: CD38 is a major NAD+ consuming enzyme whose expression and activity increase with age, accelerating NAD+ degradation. Inhibiting CD38 activity reduces NAD+ consumption and maintains intracellular NAD+ levels. Some small-molecule compounds, such as 78c and apigenin, have been reported to inhibit CD38 activity. Using CD38 inhibitors can increase NAD+ levels and improve age-related physiological dysfunction, such as enhancing cardiac function and improving metabolic disorders.
3. Lifestyle interventions: Lifestyle factors also significantly influence NAD+ levels.
Exercise: Regular exercise stimulates the NAD+ biosynthetic pathway and increases NAD+ levels. Both aerobic exercise and strength training can increase the expression and activity of NAMPT in skeletal muscle, promoting NAD+ synthesis. Exercise can also regulate the expression of NAD+ metabolism-related genes, improve mitochondrial function, and enhance cellular antioxidant capacity. In the elderly population, moderate exercise can effectively increase NAD+ content in muscles, improve muscle strength and motor function, and slow down the aging process.
Dietary restriction: Dietary restrictions, such as calorie restriction (CR) and intermittent fasting (IF), are widely recognized as effective strategies for slowing aging. These dietary patterns exert their anti-aging effects by regulating NAD+ metabolism. CR and IF activate sirtuin family proteins such as SIRT1, promoting NAD+ synthesis and utilization. Dietary restriction can also reduce oxidative stress, improve metabolic function, and reduce the risk of age-related diseases. In animal experiments, long-term calorie restriction can significantly increase NAD+ levels and extend the lifespan of multiple species.
Anti-aging Effects of Increasing NAD+ Levels
1. Anti-aging effects in animal experiments: Numerous animal experiments have confirmed that increasing NAD+ levels can significantly slow down the aging process and improve age-related physiological dysfunction.
Improved Metabolic Function: In aged mice, supplementation with NMN or NR can enhance insulin sensitivity, regulate blood glucose levels, and improve lipid metabolism disorders. NAD+ precursor supplementation can increase fatty acid oxidation in adipose tissue, reduce fat accumulation, and lower the risk of obesity-related diseases. Increasing NAD+ levels can also improve liver metabolic function, enhance the liver's detoxification capacity for drugs and toxins, and maintain normal liver physiological function.
Cardiovascular Function Protection: During the aging process, the cardiovascular system undergoes structural and functional changes, such as myocardial hypertrophy and reduced vascular elasticity. Supplementing with NAD+ precursors can improve cardiac contraction and relaxation function, reduce myocardial fibrosis, and mitigate oxidative stress damage. In animal models, supplementation with NMN or NR can lower blood pressure, improve vascular endothelial function, and reduce the risk of cardiovascular disease. In myocardial infarction models, increasing NAD+ levels can promote myocardial cell survival and repair, reduce infarct size, and improve cardiac function.
Neuroprotective Effects: In models of neurodegenerative diseases, increasing NAD+ levels demonstrates significant neuroprotective effects. Studies have shown that supplementation with NMN or NR can improve cognitive function, reduce neuroinflammation, and decrease the aggregation of neurotoxic proteins. In Alzheimer's disease mouse models, supplementation with NAD+ precursors can reduce β-amyloid production, inhibit excessive phosphorylation of tau protein, protect neurons from damage, and thereby improve learning and memory abilities.
Extended lifespan: In various model organisms, increasing NAD+ levels has been shown to extend lifespan. In nematodes and fruit flies, increasing NAD+ levels through genetic manipulation or supplementation with NAD+ precursors can significantly extend their lifespan. In mouse experiments, long-term supplementation with NMN or NR also showed a trend toward extended lifespan, although this effect may vary across different studies. Overall, these findings indicate the positive impact of increasing NAD+ levels on lifespan.
Conclusion
As an essential coenzyme within cells, NAD+ plays an indispensable role in key physiological processes such as energy metabolism, DNA repair, and post-translational modification of proteins. As age increases, the decline in NAD+ levels is closely associated with the aging process and the onset and progression of various age-related diseases. Strategies to increase NAD+ levels, such as supplementing NAD+ precursors, regulating NAD+ metabolic enzymes, and lifestyle interventions, have demonstrated significant anti-aging effects in animal experiments, including improved metabolic function, protection of the cardiovascular and nervous systems, and extended lifespan.
Sources
[1] Chubanava S, Treebak J T. Regular exercise effectively protects against the aging-associated decline in skeletal muscle NAD content[J]. Experimental Gerontology, 2023,173:112109.DOI:10.1016/j.exger.2023.112109.
[2] Soma M, Lalam S K. The role of nicotinamide mononucleotide (NMN) in anti-aging, longevity, and its potential for treating chronic conditions[J]. Molecular Biology Reports, 2022,49(10):9737-9748.DOI:10.1007/s11033-022-07459-1.
[3] Curry A, White D, Cen Y. Small Molecule Regulators Targeting NAD(+) Biosynthetic Enzymes[J]. Current Medicinal Chemistry, 2022,29(10):1718-1738.DOI:10.2174/0929867328666210531144629.
[4] Yuan Y, Liang B, Liu X, et al. Targeting NAD+: is it a common strategy to delay heart aging?[J]. Cell Death Discovery, 2022,8. https://api.semanticscholar.org/CorpusID:248393418
[5] Levine D C, Hong H, Weidemann B J, et al. NAD(+) Controls Circadian Reprogramming through PER2 Nuclear Translocation to Counter Aging[J]. Molecular Cell, 2020,78(5):835-849.DOI:10.1016/j.molcel.2020.04.010.
[6] Fang E F, Hou Y, Lautrup S, et al. NAD(+) augmentation restores mitophagy and limits accelerated aging in Werner syndrome[J]. Nature Communications, 2019,10(1):5284.DOI:10.1038/s41467-019-13172-8.
[7] Yaku K, Okabe K, Nakagawa T. NAD metabolism: Implications in aging and longevity[J]. Ageing Research Reviews, 2018,47:1-17.DOI:10.1016/j.arr.2018.05.006.
[8] Chaturvedi P, Tyagi S C. NAD(+) : A big player in cardiac and skeletal muscle remodeling and aging[J]. Journal of Cellular Physiology, 2018,233(3):1895-1896.DOI:10.1002/jcp.26014.
Product available for research use only:
![2 2]()
English
العربية
Français
Русский
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy